And don’t forget to put the unit g/mol to your final calculated molar mass.First solve the brackets, then multiplications and at last do the final addition. 1 mole of a substance contains 6.022 x 10 23 molecules. Molecular mass of nitrogen (N 2) 14 x 2 28 g. Always follow the calculation order to avoid any mistakes in calculation. mass of one nitrogen atom and nitrogen molecule in kg.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole.I hope you have understood the short and simple calculation for finding the molar mass of N2. 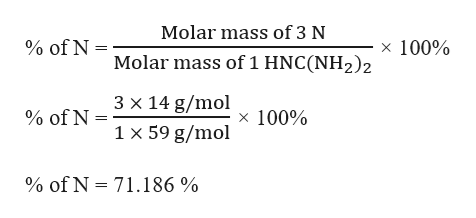
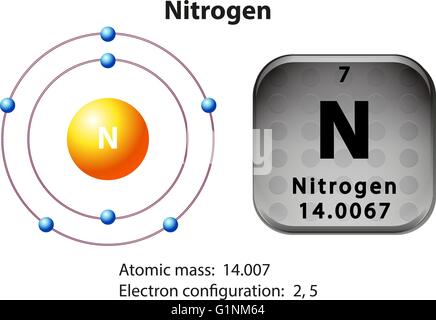
Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and. Hence the Molar mass of N2 is 28.014 g/mol. Element Nitrogen (N), Group 15, Atomic Number 7, p-block, Mass 14.007. A liquid nitrogen shower (-321F) locks in the. To understand whether the process of seasonal nitrogen resorption and biomass allocation are different in CO2-enriched plants, seedlings of red maple (Acer. So, Molar mass of N2 = Molar mass of 2 Nitrogen (N) atoms. The result perfectly brewed coffee at 10x strength is flash-frozen moments later. You can see that in N2, there are 2 Nitrogen atoms. The plants provide shelter and even a few essential nutrients, jealously guarding their guests, and the bacteria provide a steady supply of nitrogen.Now, to calculate the molar mass of N2, you just have to add the molar mass of all the individual atoms that are present in N2. For instance, legumes build special nodules in their roots that provide a perfect home for the bacteria. IN your case, nitrogen gas has a molar mass of M M 28.0134 g mol1 This means that every mole of nitrogen gas has a mass of 28.0134 g. The particulate organic carbon (POC), particulate organic nitrogen (PON), dry mass, and chlorophyll a content of marine snow as a function of individual. Fortunately, nitrogen-fixing bacteria are found throughout the world, and are often found in partnerships with plants. This is a large investment in energy, but well worth the effort if nitrogen is not available in the environment. For all source categories, NOx is compiled from emissions measurements that express NOx mass based on the molecular weight of nitrogen dioxide (NO2). Nitrogenase also converts hydrogen ions to hydrogen gas at the same time (this might be an obligatory part of the nitrogen-splitting reaction, or it might be a simple side effect), thus consuming even more ATP in the process. In the typical reaction, two molecules of ATP are consumed for each electron transferred. The Fe protein, shown in green, uses the breakage of ATP to pump these electrons into the MoFe protein. The reaction requires the addition of six electrons for each nitrogen molecule that is split into two ammonia molecules. No matter what operating system you are using, mMass works on MS Windows, Apple's Mac OS X and Linux platforms as well. Dry mass 4.90 0.1 ( n 6 ) 4.66 0.1 ( n 6 ) Nitrogen content of dry. The present study applies for the first time as Matrix-Assisted Laser Desorption/Ionization (MALDI) Mass Spectrometry Imaging (MSI) on real thyroid Fine Needle Aspirations (FNAs) to test its possible complementary role in routine cytology in the diagnosis of thyroid nodules. Stay focused on your data interpretation, not on the software. Nitrogen biomass up - take for systems fed with de - gritted wastewater and. mMass is designed to be feature rich, yet still easy to use. The MoFe protein, shown in blue and purple, contains all of the machinery to perform the reaction, but requires a steady source of electrons. mMass - Open Source Mass Spectrometry Tool. This is a DOS application no other software is required except a 'zip' utility to extract the files. So, a) the mass of 1 mole of nitrogen 1 amu in gram 1 14 14 gm. Atomic mass in gram represents one mole of that substance. What is the mass of 1 mole of nitrogen atoms Studdy. We know that one mole of a particle will contain Avogadro number of atoms. Minimum 640K RAM is needed with 350K resident memory storage. The mass of one mole of nitrogen atom will be 14,g. 
Nitrogenase is composed of two components, shown here from entry 1n2c. The user-friendly model, computerized by our contractor, Horsley and Witten, Inc., is stand-alone software originally written for IBM-compatible computers operating on a DOS system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
